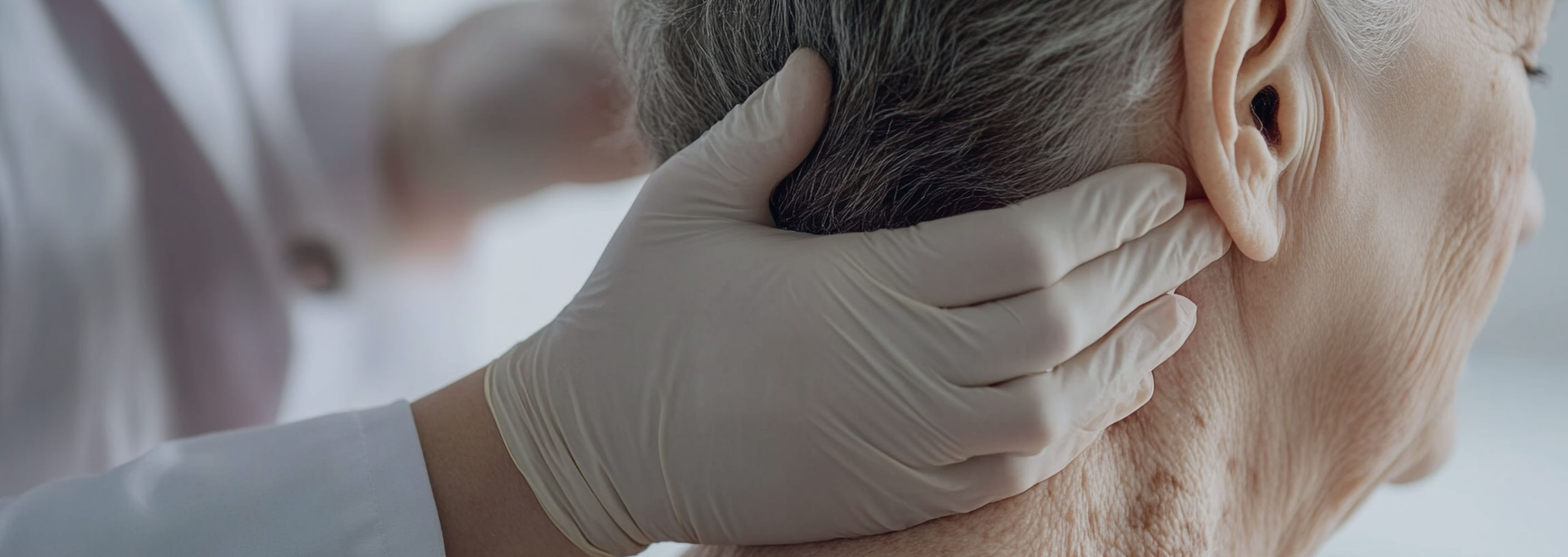
Research and Development
Biofrontera Inc. is conducting extensive research to optimize the market potential of AMELUZ® and BF-RhodoLED®.
Biofrontera Inc. has a wholly-owned subsidiary, Biofrontera Discovery, which is based in Germany and represents the scientific and operational backbone of the company. Biofrontera Discovery assumes significant global functions that are essential to the company’s sustainable growth and long-term leadership in Photodynamic Therapy, which include clinical development, regulatory affairs, quality and pharmacovigilance and manufacturing.
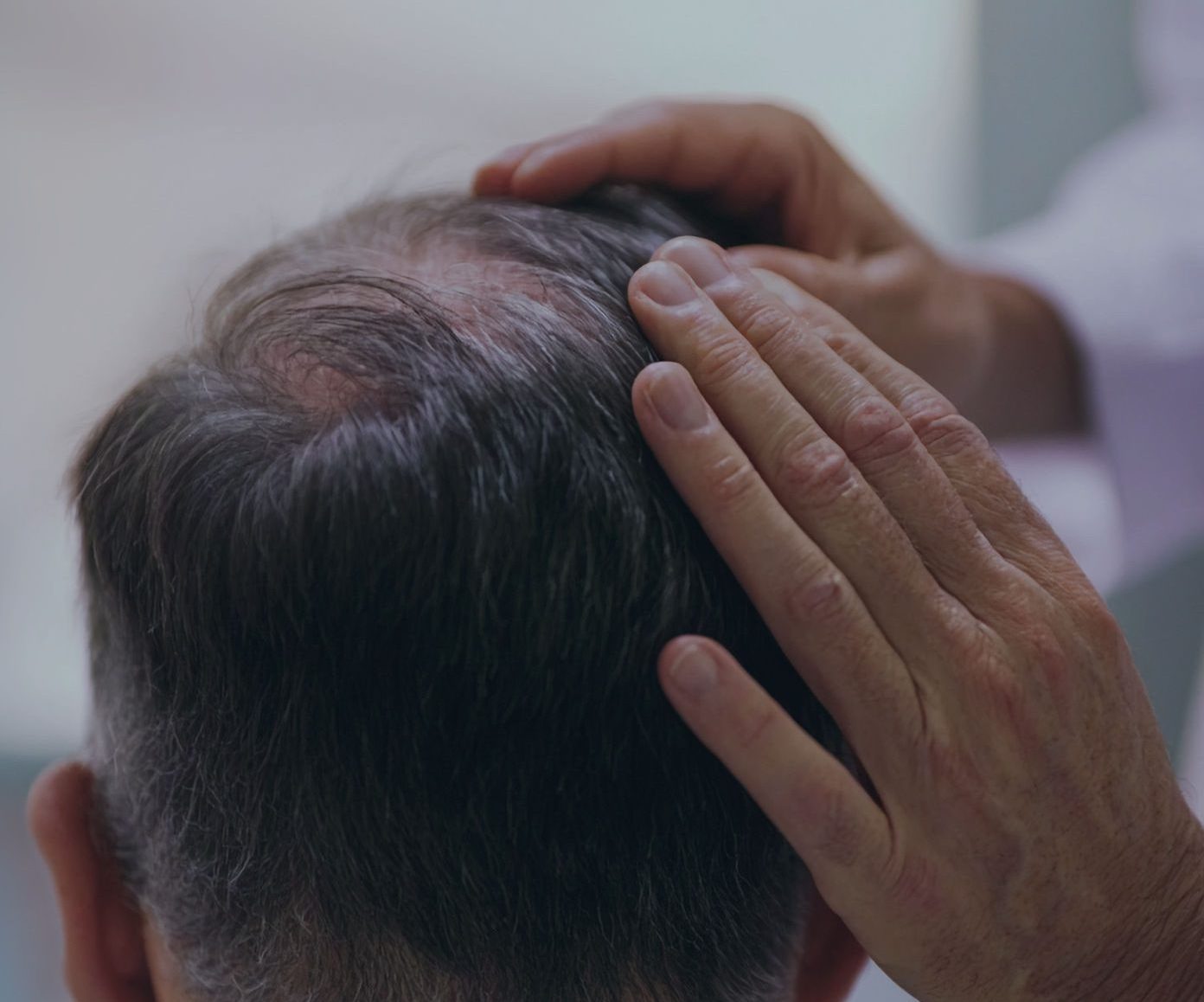
This clinical development includes both late-stage and early-stage product candidates targeting specific treatment needs in the dermatology market. For over a decade, Biofrontera has deepened its expertise in dermatology by advancing photodynamic therapy (PDT) research, ultimately expanding treatment accessibility and helping healthcare professionals improve patient lives.
Product & Indication
RhodoLED® XL
Mild to Moderate Actinic Keratosis: Face & Scalp, in combination with AMELUZ®
AMELUZ®
New Formulation
AMELUZ®
Mild to Moderate Actinic Keratosis: Face & Scalp Larger Tx Area (3 tubes)
AMELUZ®
Superficial Basal Cell Carcinoma
AMELUZ®
Actinic Keratosis: Trunk & Extremities
AMELUZ®
Moderate to Severe Acne Vulgaris
AMELUZ®
Actinic Keratosis: Larger Tx Area PK
